Precision medicine is changing how clinicians think about bronchiectasis. For years, treatment has relied on broad labels such as symptom burden, exacerbation history, sputum culture results, and whether Pseudomonas aeruginosa is present. That approach is still relevant, but it does not fully explain a frustrating reality seen in daily practice and in clinical trials: inhaled antibiotics can work very well in some people, yet offer modest or inconsistent benefit in others. Recent research suggests the answer may lie in biologically distinct subgroups, or endotypes, rather than in standard clinical labels alone.
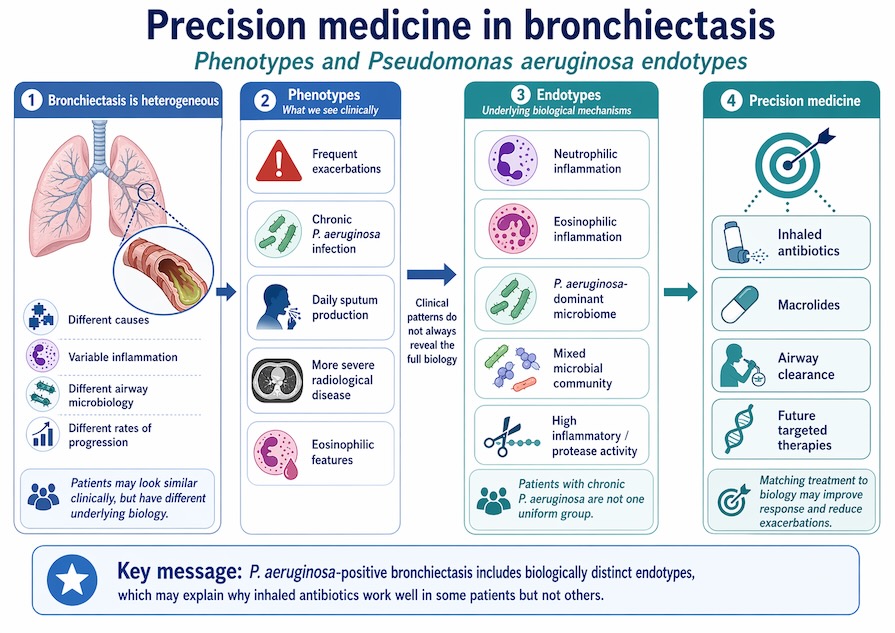
Bronchiectasis is not a single disease. It is the final result of many different insults. It is also a syndrome with many causes, inflammatory patterns, airway microbiology profiles, and rates of progression. Two patients can both have chronic cough, daily sputum, repeated chest infections, and positive cultures for Pseudomonas aeruginosa, yet still behave very differently over time. One may improve clearly on inhaled antibiotics, with fewer flare-ups and better quality of life. Another may continue to exacerbate despite apparently appropriate treatment. This heterogeneity is one of the main reasons precision medicine has become such an important goal in bronchiectasis research.
Current guidelines still support inhaled antibiotics for selected patients, especially those with chronic Pseudomonas aeruginosa infection and frequent exacerbations. The 2025 European Respiratory Society guideline recommends offering long-term inhaled antibiotics to adults at high risk of exacerbations who have chronic Pseudomonas aeruginosa infection despite standard care. Older but still influential British Thoracic Society guidance similarly recommends inhaled colistin for chronic Pseudomonas infection and considers inhaled gentamicin as an alternative, with long-term antibiotics generally considered in people with three or more exacerbations a year. NICE’s current primary care guidance also reflects this specialist-led approach.
Even so, the clinical trial record for inhaled antibiotics in bronchiectasis has often looked mixed. A 2024 systematic review and meta-analysis of 20 studies involving 3,468 patients found that inhaled antibiotics produced a slight reduction in exacerbations, a probable reduction in severe exacerbations, and a likely slight improvement in symptoms. That is encouraging, but it also shows the average effect is not dramatic for every patient. Earlier trial programmes, including ORBIT and RESPIRE, also produced results that were promising in places but inconsistent across studies and endpoints. This pattern has pushed researchers to ask a better question: not simply “Do inhaled antibiotics work?” but “Who exactly are they working for?”
That is where endotypes come in. A phenotype describes what a patient looks like clinically, such as frequent exacerbations, chronic infection, or radiological severity. An endotype goes deeper and describes the biological mechanisms driving disease in that subgroup. In bronchiectasis, researchers are increasingly trying to separate patients by inflammatory pathways, microbiome structure, immune response, and molecular signatures. The aim is to match the right therapy to the right biology instead of treating all patients with the same label in the same way.
The most important recent advance in this area is the 2025 work on Pseudomonas aeruginosa endotypes using samples from the ORBIT-3 and ORBIT-4 phase III trials of inhaled liposomal ciprofloxacin. These trials originally appeared inconsistent, with one showing clearer benefit than the other. The newer analysis looked beyond routine clinical characteristics and examined sputum microbiota, inflammatory markers, proteomics, and geographical variation. The conclusion was striking: patients with chronic Pseudomonas aeruginosa infection do not form one uniform group. They show heterogeneous microbiota and inflammatory profiles, and those differences are linked to both exacerbation frequency and response to inhaled antibiotic treatment.
This is important because it may explain why repeat trials of seemingly similar patients can produce different headline results. In the new analysis, once the researchers adjusted for microbiota profile and region, the treatment estimates in ORBIT-3 and ORBIT-4 became much more similar and aligned with prior meta-analyses of inhaled antibiotics in bronchiectasis. In practical terms, that suggests some past inconsistency was not random noise. It may have reflected meaningful biological differences between enrolled patient groups. In other words, “chronic Pseudomonas infection” may be too broad a category for precision treatment decisions and even for trial design.
For clinicians, this is an important shift. Until now, sputum culture has been the main microbial guide to treatment. Culture remains valuable, but it offers only a partial view. Two sputum samples can both grow Pseudomonas aeruginosa while hiding major differences in bacterial community structure, total bacterial load, inflammatory activity, and host response. Precision medicine suggests that these hidden differences may determine whether inhaled antibiotics reduce flare-ups, improve symptoms, or make little difference. This is a more biologically realistic way to think about response.
It also fits with the wider direction of bronchiectasis research. Recent reviews describe several candidate endotypes beyond chronic Pseudomonas infection alone. These include neutrophilic, eosinophilic, mixed inflammatory, infective, exacerbation-prone, and immune-related patterns. Some studies now suggest that eosinophilic bronchiectasis could represent a clinically relevant subgroup, while other work has explored molecular signatures linked to antibiotic response. The field is still early, but the broader message is clear: bronchiectasis biology is more diverse than traditional categories suggest.
For patients, none of this means inhaled antibiotics are suddenly obsolete or only suitable after complex omics testing. Inhaled antibiotics remain a sensible and guideline-supported option in the right setting, particularly for people with chronic Pseudomonas infection and repeated exacerbations. What is changing is the expectation that future care will become more targeted. Rather than asking every patient with Pseudomonas to try the same treatment pathway, specialists may increasingly use biomarkers, sputum profiling, inflammatory measures, and clinical history to predict who is most likely to benefit and who might need another strategy.
This could improve care in several ways. First, it may spare some patients months of treatment burden from inhaled therapy that is unlikely to help them. Second, it may help identify people who need combination approaches, such as inhaled antibiotics plus macrolides, aggressive airway clearance, or future biologically targeted therapies. Third, it could make clinical trials more reliable by enrolling patients whose disease biology matches the drug being tested. That would be especially important in bronchiectasis, where failed trials may sometimes reflect poor patient stratification rather than a truly ineffective treatment.
The implications reach beyond antibiotics. Precision medicine in bronchiectasis is also opening the door to non-antibiotic approaches aimed at specific inflammatory or microbial pathways. A recent University of Dundee report on gremubamab, an anti-Pseudomonas monoclonal antibody approach, described encouraging early findings and highlighted the lack of approved therapies that specifically target bronchiectasis or Pseudomonas aeruginosa. While this work is still developing, it shows how endotyping could eventually support a broader treatment model, where infection-directed antibodies, anti-inflammatory drugs, and inhaled antibiotics are matched to the biology of each patient.
There are still important limits. Endotyping tools are not yet routine in most clinics. Many studies remain exploratory, and some rely on advanced molecular methods that are not widely available. Biomarkers also need to be reproducible, affordable, and clinically useful before they can guide standard care. In addition, bronchiectasis management will always depend on fundamentals that precision medicine cannot replace: airway clearance, exercise, vaccination, smoking cessation, treatment of underlying causes, and careful review of exacerbation history and microbiology.
For the next few years, the most likely outcome is not a sudden revolution but a gradual change in practice. Sputum cultures will remain important, but it may be joined by better biomarker panels. Trial populations may become more tightly defined. Clinicians may start to think of chronic Pseudomonas infection less as a single end-stage label and more as a family of biological states. As that happens, inhaled antibiotics may finally find a clearer place: not as treatments that work inconsistently in a broad disease, but as therapies that work predictably in the right endotype.
FAQs
What is precision medicine in bronchiectasis?
Precision medicine in bronchiectasis means tailoring treatment to the patient’s underlying biology, not just their symptoms or sputum culture. It aims to identify which subgroup, or endotype, a patient belongs to and then match therapy more accurately.
Why do inhaled antibiotics help some bronchiectasis patients more than others?
Recent research suggests response may depend on differences in airway microbiota, inflammatory activity, and host biology, even among patients who all culture positive for Pseudomonas aeruginosa.
What are Pseudomonas endotypes?
These are biologically distinct subgroups among patients with chronic Pseudomonas aeruginosa infection. They are defined by different microbial and inflammatory patterns and may be linked to different treatment responses.
Are inhaled antibiotics still recommended now?
Yes. Current guidelines continue to recommend long-term inhaled antibiotics for selected patients, especially those with chronic Pseudomonas infection and frequent exacerbations, usually under specialist care.
Will endotyping soon become routine in clinic?
Not yet. The science is advancing quickly, but most endotyping approaches still need wider validation and practical tools before they become standard in everyday bronchiectasis care.
GET IN TOUCH
Schedule a Visit with Dr Jose
For an assessment of your diagnosis and treatment.
Disclaimer: The information provided in this article is for informational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment, and is not an advertisement for medical products. Always seek the advice of your healthcare provider with any questions you may have regarding a medical condition or treatment. Your healthcare professional can assess your individual circumstances. Consultation does not guarantee suitability for any specific treatment; all clinical decisions follow an individual assessment and shared decision-making
